Furthermore, CSP is used in the prediction of stoichiometric hydrates 34 and solvates 35, 36, but without explicitly considering relative humidity or solvent activity. CSP can be used to identify the most stable, possibly still to be discovered, crystal form of pharmaceutically relevant molecules 20, 21, 32 and has started to be applied early in the molecular and materials design cycles by balancing accuracy and computational efficiency 33. The capabilities of CSP have improved greatly in recent years by the inclusion of temperature-dependent free-energy calculations, yet without rigorously assessing the accuracy of such predictions 2, 4, 24, 25, 26, 27, 28, 29, 30, 31. To complement experimental efforts, computational methods, in particular crystal structure prediction (CSP), are becoming important for polymorph risk assessment and control 3, 5, 20, 21, 22, 23. 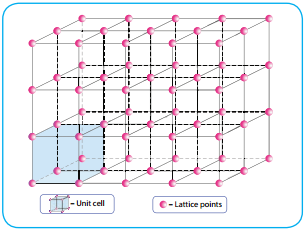
Crystal form selection remains a challenge because of the implications for physical and chemical stability, solubility, dissolution, nucleation barriers, mechanical properties, filtration, powder flow and, under consideration here, the formation of hydrates and solvates stable within accessible temperature and humidity ranges 1, 17, 18, 19. The development of an undesirable crystal form can have harmful consequences, as shown in the cases of ritonavir and rotigotine 12, 15, 16. Their physicochemical properties depend on an interplay of chemical composition and molecular packing within a crystal structure, known as a crystal form or polymorph when more than one arrangement exists 10, 11, 13, 14. Molecular crystals are important components of food products 6, semiconductors 7, explosives 8, agrochemicals 9 and pharmaceuticals 10, 11, 12. These contributions reduce the gap between the needs of the experimentalist and the capabilities of modern computational tools, transforming crystal structure prediction into a more reliable and actionable procedure that can be used in combination with experimental evidence to direct crystal form selection and establish control 5. The calculated free energies have standard errors of 1–2 kJ mol −1 for industrially relevant compounds, and the method to place crystal structures with different hydrate stoichiometries on the same energy landscape can be extended to other multi-component systems, including solvates. Here we redefine the state of the art, primarily by improving the accuracy of free-energy calculations, constructing a reliable experimental benchmark for solid–solid free-energy differences, quantifying statistical errors for the computed free energies and placing both hydrate crystal structures of different stoichiometries and anhydrate crystal structures on the same energy landscape, with defined error bars, as a function of temperature and relative humidity. In silico crystal form selection has recently come much closer to realization because of the development of accurate and affordable free-energy calculations 2, 3, 4. The physicochemical properties of molecular crystals, such as solubility, stability, compactability, melting behaviour and bioavailability, depend on their crystal form 1. Nature volume 623, pages 324–328 ( 2023) Cite this article \): The Three Kinds of Cubic Unit Cell.Predicting crystal form stability under real-world conditions
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
